When a doctor prescribes a medication like vancomycin, lithium, or tacrolimus, they're not just giving you a pill-they're relying on precise lab results to keep you safe. These drugs have a therapeutic window: too little won't work, too much can kill you. That’s why confirming the lab’s monitoring process isn’t optional-it’s life-or-death. Yet, most patients never ask how the lab ensures those numbers are accurate. Here’s how it actually works behind the scenes.
Verification vs. Validation: What’s the Difference?
Not all lab tests are created equal. If a test is FDA-cleared (like most common drug level checks), the lab does verification. That means they check if the test performs exactly as the manufacturer claims. For example, if Abbott says their lithium test has a precision of ±5%, the lab runs 20 samples over five days to prove it. No guesswork. Just data. But if the drug is something rare-like a metabolite of clozapine-there’s no FDA-approved test. Then the lab must do validation. This is a full rebuild: they have to prove accuracy, sensitivity, specificity, and linearity from scratch. Validation takes three times longer, costs up to three times more, and fails nearly 20% of the time. That’s why many hospitals avoid validating new tests unless absolutely necessary.The 9-Step Process No One Talks About
Every time a lab adds a new medication test, they follow a strict 9-step protocol. Skipping even one step risks patient safety.- Write a plan: Define exactly what performance standards the test must meet.
- Get lab director approval: No test runs without a pathologist’s signature.
- Run the tests: At least 20 replicates over five days to check precision.
- Analyze the numbers: Is the bias under half the allowable error? Is linearity R² ≥0.99?
- Re-test if needed: About 30% of tests need tweaks before approval.
- Write the summary report: Every number, every outlier, every deviation documented.
- Create SOPs and training: Staff must know exactly how to run it-and when to flag a problem.
- Final approval: Another sign-off from the director.
- Go live: The test is now active for patient samples.
This process takes 80-120 hours per test. For a busy lab adding three new drugs? That’s over 300 hours of work-often done by one overworked quality specialist.
What Can Go Wrong? Real Examples
In 2021, a hospital in Ohio mis-dosed 23 patients because their digoxin test didn’t account for antibody interference. The lab never tested for cross-reactivity with common drugs like quinidine. That’s not negligence-it’s a failure to follow CLSI EP15-A3 guidelines. Another case: a lab verified a tacrolimus test but skipped checking metabolite interference. Three transplant patients had falsely low results. They were given higher doses, leading to kidney damage. The lab later admitted they didn’t have access to clinical samples with high metabolite levels. That’s a common problem-only 59% of labs can get enough real-world samples to properly validate tests.The Hidden Gaps in Today’s Labs
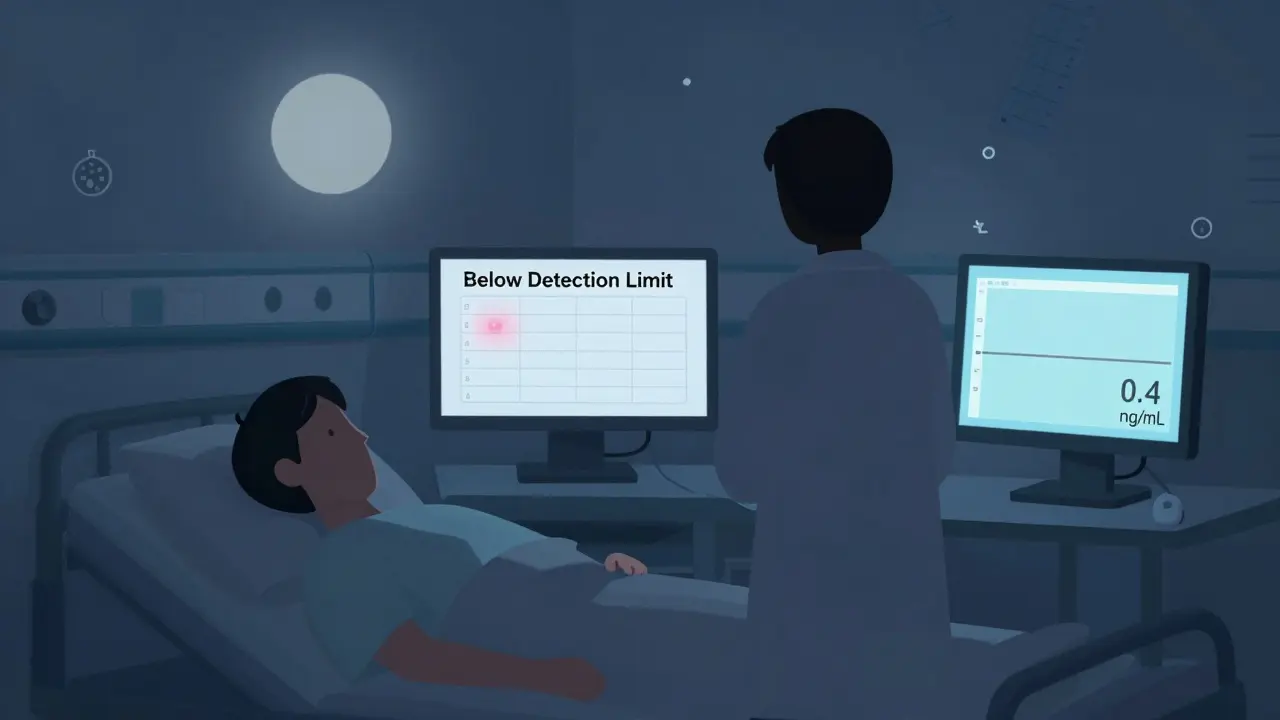
A 2023 CAP survey found only 64% of labs verify the lower limit of quantitation for narrow therapeutic index drugs. That’s terrifying. If a patient’s level is 0.4 ng/mL and the test can’t reliably detect below 0.6 ng/mL, the lab reports “below detection limit.” The doctor assumes it’s safe. But it’s not. It’s just invisible. Another issue? Manufacturer claims. Abbott scores 4.7/5 for clear documentation. Smaller manufacturers? 3.2/5. One lab director told me: “I spent 11 hours trying to find the interference profile for a new antiepileptic test. The manual didn’t even list the top three drugs that interfere.”What Patients Should Ask Their Provider
You don’t need to understand statistical linearity. But you do need to know this:- Ask if your medication is monitored with an FDA-cleared test.
- If it’s a newer drug (like a biologic or targeted cancer therapy), ask if the lab validated the test.
- Request a copy of your lab report. Look for “result below limit of quantitation.” If it’s there, ask what the actual number was.
- If your dose keeps changing without clear reason, ask if the lab recently changed its assay.
Most providers don’t know the details either. But if you ask, it forces them to check. And that’s the point.
Why This Matters More Than Ever
The global therapeutic drug monitoring market is growing at 7.8% per year. More drugs now require precise dosing-especially immunosuppressants, anticoagulants, and psychiatric meds. But staffing is collapsing. 68% of lab directors say they can’t hire enough qualified staff to keep up with verification demands. That means corners get cut. A lab might skip interference testing because they’re short-staffed. Or they might use an old SOP from 2018 for a new test. That’s how errors creep in. The FDA’s new 2024 draft guidance now requires testing against 15 metabolites for immunosuppressants-not just 8. Labs that haven’t updated their protocols are already out of compliance. And under CLIA, non-compliance costs $7,500 per violation. But the real cost? A patient’s life.What’s Changing in 2025
The CLSI is finalizing EP44-Ed2, a new standard for narrow therapeutic index drugs. It will require stricter verification for all drugs with a therapeutic window under ±10%-including digoxin, warfarin, and phenytoin. EHRs are starting to help. Epic Systems now flags sub-therapeutic levels automatically in 45% of US hospitals. If your vancomycin level drops to 8 mcg/mL (below the 15-20 range), the system sends an alert to your doctor before you even leave the clinic. But technology alone won’t fix this. The human process-verification, documentation, training-still matters most. A 2022 study found labs following ISO 15189 had 47% fewer medication errors. That’s not because they had better machines. It’s because they had better systems.Final Thought: Trust But Verify
Your doctor trusts the lab. The lab trusts the manufacturer. The manufacturer trusts their own data. And somewhere in that chain, a mistake can happen. The system works-but only if every link is checked. That’s why confirmation isn’t just a lab procedure. It’s a patient safety practice. And if you’re on a medication that needs monitoring, you have a right to know it’s being done right.What’s the difference between verification and validation in lab testing?
Verification is used for FDA-cleared or approved tests and confirms the test performs as the manufacturer claims. Validation is for laboratory-developed tests (LDTs) or modified tests and requires full performance testing from scratch. Verification takes 80 hours on average; validation takes 120+ hours and costs 2-3 times more.
Which medications require the strictest lab monitoring?
Drugs with a narrow therapeutic index need the strictest monitoring. These include vancomycin (target range: 15-20 mcg/mL), digoxin (0.5-4.0 ng/mL), lithium (0.6-1.2 mmol/L), and tacrolimus (5-15 ng/mL). Even small errors in measurement can lead to toxicity or treatment failure.
Why is interference testing so important?
Interference occurs when other substances in the blood-like metabolites or other drugs-trick the test into giving a false result. For example, carbamazepine metabolites can cause falsely high levels in immunoassays. Without testing for interference, labs can miss this, leading to dangerous overdoses. The most common verification failure is skipping this step.
Can a lab report a result below the detection limit?
Yes, but it’s risky. If the lower limit of quantitation isn’t properly verified, a result of “below detection limit” might actually be a sub-therapeutic level. For example, if the limit is set at 0.6 ng/mL but the therapeutic range starts at 0.5 ng/mL, the lab may miss patients who need a dose increase. Only 64% of labs verify this critical threshold.
How can I check if my lab follows proper protocols?
Ask your provider if the test is FDA-cleared or lab-developed. Request a copy of your lab report and look for the method used. If the test was recently changed, ask if the lab re-verified it. Hospitals accredited by CAP or following ISO 15189 are more likely to have robust verification. You can also ask if the lab participates in proficiency testing programs like CAP’s TDM survey.



Kevin Y.
March 26, 2026 AT 01:17Really well-explained breakdown. I work in hospital admin and see how often verification gets pushed aside when budgets are tight. It’s not that anyone’s lazy-it’s that the system doesn’t reward transparency. We just got audited last quarter, and I swear, half the team didn’t even know what CLSI EP15-A3 was. Scary stuff.
Caroline Bonner
March 27, 2026 AT 01:39I just want to say-this is the kind of content that makes me feel less alone in this world. As a nurse who’s had to double-check labs every single shift because I didn’t trust the numbers, I’ve spent years quietly freaking out. Thank you for putting this out there. The 9-step process? I’ve seen step 3 skipped. Twice. And both times, someone almost died. Please, if you’re on lithium or vancomycin-ask. Ask. ASK. Your life might depend on it.
Zola Parker
March 28, 2026 AT 06:28Wow. So the whole system is just… a trust fall? 😅 I mean, if a lab can’t even verify the lower limit of quantitation, then what’s the point of having a number at all? Are we just playing roulette with our meds now? 🎰
Linda Foster
March 29, 2026 AT 18:37Thank you for this comprehensive overview. The distinction between verification and validation is critical, and too often conflated. I would add that proficiency testing programs, such as those offered by CAP, are not optional-they are the minimum standard. Labs that do not participate in these programs should not be considered reliable for therapeutic drug monitoring.
Rama Rish
March 31, 2026 AT 06:15so if ur on tacrolimus and ur doc says ur levels are fine but u still feel awful… ask if the test was validated. not just verified. its a big deal.
Kevin Siewe
April 1, 2026 AT 03:12This is one of those posts that should be shared with every patient on immunosuppressants. I’ve seen too many people get discharged with a ‘normal’ tacrolimus level, only to crash a week later because the lab didn’t account for metabolites. Don’t assume. Ask. And if they brush you off? Get a second opinion. Your body deserves better.
James Moreau
April 2, 2026 AT 17:36As someone who grew up in a country where labs don’t even have basic QA, this was eye-opening. I didn’t realize how much structure exists here in the US. But even here, it’s fragile. We need more public awareness-not just for patients, but for policymakers. This isn’t just medical. It’s ethical.
Seth Eugenne
April 3, 2026 AT 12:07Just had my vancomycin level checked last week. Saw the note: 'below limit of quantitation.' I asked for the actual number. They gave me 12.3 mcg/mL. I was told it was 'fine.' But the range is 15–20. 😅 Thanks to this post, I’m following up with my nephrologist. You saved me from a bad decision. 🙏
Brandon Shatley
April 3, 2026 AT 19:54so i read this and my brain just went… wait so if the test says my drug level is 0.4 but the limit is 0.6… does that mean i might be getting too much? or too little? i dont even know anymore. im gonna call my dr tomorrow. lol
Rachele Tycksen
April 5, 2026 AT 17:55cool post. i didn’t know labs had to do all this. guess i’ll start asking my doc if they ‘verified’ my meds. lol
Aaron Sims
April 5, 2026 AT 23:35Of course the labs are cutting corners. 🤡 Why? Because the FDA is a puppet of Big Pharma. They want you dependent on meds. They don’t care if your lithium level is 0.6 or 1.8-just as long as you keep paying. And guess what? The ‘9-step process’? It’s a show. You think they actually run 20 replicates? Nah. They run one. Print the result. Move on. 💀
Anil Arekar
April 7, 2026 AT 11:41Thank you for sharing this detailed insight. In my country, access to such rigorous testing is rare. But the principles here are universal. Patient safety must not be a luxury. I hope this becomes a global standard, not just an American one. We must elevate lab science to the dignity it deserves.
Chris Crosson
April 9, 2026 AT 02:52Can someone explain why the FDA doesn’t mandate full validation for all narrow therapeutic index drugs? If the risk is life-or-death, why is verification enough? This feels like a regulatory loophole that’s killing people. We need legislation. Not just awareness.
Katie Putbrese
April 10, 2026 AT 18:05This is why America is falling apart. Labs are run by overpaid bureaucrats who think ‘R² ≥0.99’ means something. Meanwhile, real doctors are drowning in paperwork. We need to cut the red tape, not add more steps. Trust your doctor. Not some spreadsheet. 🇺🇸
Kevin Y.
April 11, 2026 AT 06:23Just saw this reply and had to respond. You’re right to be frustrated-but cutting red tape won’t fix this. The 9-step process exists because people died. We don’t need less oversight. We need better funding. And more trained staff. The real villain here isn’t bureaucracy-it’s underinvestment.