Imagine waking up and having your tremors suddenly vanish, or finding that the "off" periods where your medication stops working have shrunk from six hours to just one. For many people living with Parkinson's, this isn't a dream-it's the result of Parkinson’s DBS is a neurosurgical procedure that uses implanted electrodes to regulate abnormal brain activity, effectively acting as a pacemaker for the brain. While it sounds like science fiction, it's a practical tool used to reclaim independence when medications like levodopa start to fail. However, the biggest hurdle isn't the surgery itself; it's figuring out if you're actually a good fit for it.
The Bottom Line on DBS
Before getting into the weeds, here is the quick version: DBS doesn't cure Parkinson's or stop the disease from progressing. Instead, it manages the symptoms. If your meds used to work great but now cause uncontrollable jerky movements (dyskinesia) or leave you frozen for hours, you might be a candidate. The goal is to reduce the "rollercoaster" effect of medication and give you more stable, fluid movement throughout the day.
| Symptom | Typical Improvement | Impact on Daily Life |
|---|---|---|
| Motor Fluctuations | 60-80% reduction | Less "off" time; more predictable movement. |
| Dyskinesias | Up to 80% reduction | Fewer involuntary twisting movements. |
| Medication Load | 30-50% reduction | Lower doses of levodopa; fewer drug side effects. |
How the Technology Actually Works
Think of Deep Brain Stimulation as a way to "jam" the wrong signals in the brain. In Parkinson's, certain areas of the brain fire off electrical signals in an erratic way, which leads to tremors and stiffness. Surgeons implant thin electrodes-only about 1.27mm wide-into specific targets like the Subthalamic Nucleus (STN) or the Globus Pallidus interna (GPi). These wires connect to an Implantable Pulse Generator (IPG), a small battery pack usually tucked under the collarbone.
Modern systems, such as the Medtronic Percept PC or Boston Scientific Vercise Genus, are far more advanced than the early models from the 90s. We now have "directional leads" that let doctors steer the electricity more precisely, and "closed-loop" technology. Closed-loop systems are a game changer; they actually sense your brain's beta band oscillations (13-35 Hz) and adjust the stimulation in real-time. This means the device gives you more power when you're stiff and backs off when you don't need it, which can lead to 27% better symptom control compared to old-school "constant" stimulation.
Choosing the Right Target: STN vs GPi
One of the biggest decisions during candidate selection is where to put the electrodes. It’s not a one-size-fits-all choice. The STN is the most popular target because it allows patients to significantly drop their medication dosage. However, some people find it can cause slight issues with word-finding or cognitive speed. On the other hand, the GPi is often the go-to for people whose main struggle is severe dyskinesia. It tends to be gentler on the brain's cognitive functions, though you might not be able to cut your meds as drastically.
Which one wins? According to the VA/NINDS CSP #468 trial, both targets provide roughly the same overall motor improvement (about 49%). The choice really comes down to your specific symptoms: if you want to get off meds, go STN; if you want to stop the twisting movements and protect your cognition, GPi is often the better bet.
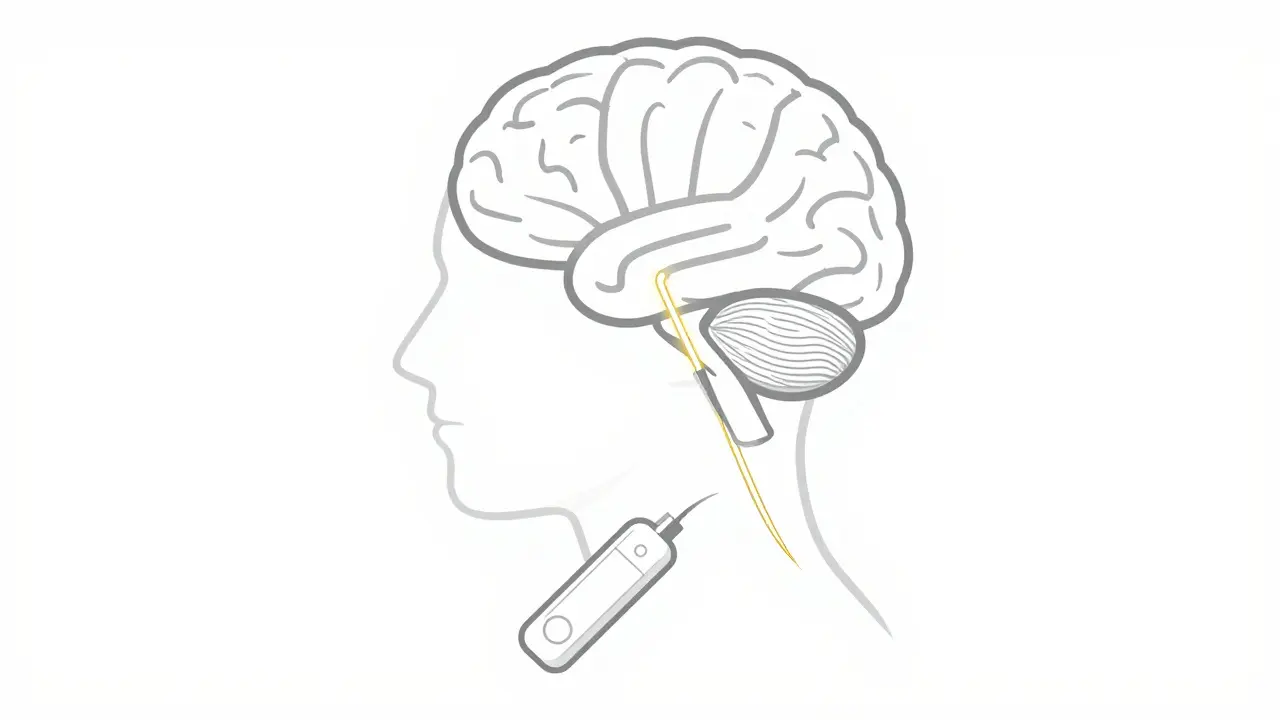
Are You a Candidate? The Selection Process
You can't just walk into a clinic and request DBS. There is a rigorous 3-6 month vetting process to ensure you'll actually benefit. The most critical rule of thumb is the "levodopa response." If a dose of levodopa doesn't improve your motor scores by at least 30%, DBS likely won't either. Why? Because DBS essentially mimics the effect of the drug. If the drug doesn't work, the electricity won't work.
Beyond the motor side, doctors look at a few other key markers:
- Disease Duration: Usually, a minimum of 5 years with the diagnosis is required to ensure the symptoms are stable and not due to another condition.
- Cognitive Health: This is a deal-breaker. If you have significant dementia or a low score on a MoCA or MMSE test, DBS is usually avoided. The surgery can actually worsen cognitive decline in these cases.
- Mental Health: Severe, untreated depression or anxiety can be exacerbated by the procedure.
- Physical Health: Since this involves brain surgery, your heart and general health must be stable enough to handle local anesthesia and sedation.
It's also worth noting that DBS is for idiopathic Parkinson's. If you have "Parkinson-plus" syndromes-like Multiple System Atrophy (MSA) or Progressive Supranuclear Palsy (PSP)-the response rate drops below 10%. For these conditions, the surgery is generally not recommended.
The Real-World Experience: Pros and Cons
If you talk to people in the Parkinson's community, you'll see that the results are usually life-changing, but they aren't perfect. Many report a massive drop in "off" time. One patient noted their off-time went from six hours a day to just one. That's the difference between being bedridden and being able to go for a walk.
However, there are trade-offs. First, the learning curve. You don't just wake up from surgery and feel great. The programming phase takes 6 to 12 months of fine-tuning. You'll visit your neurologist frequently to adjust voltages and frequencies until the "sweet spot" is found. Then there's the hardware. Even with rechargeable batteries that last 9-15 years, some people still face hardware complications or infections (about 5-15% of cases).
And here is the most important reality check: DBS doesn't fix everything. Axial symptoms-like balance, gait, and swallowing-often show only 20-30% improvement. If your main problem is falling over or a frozen gait, don't expect DBS to be a magic wand. It fixes the "shaking and stiffness" far better than it fixes the "balance and walking."

Comparing DBS to Other Options
You might hear about Focused Ultrasound. This is a non-invasive alternative that uses sound waves to create a tiny lesion in the brain. It's great because there's no surgery and no hardware. The catch? It's usually only done on one side of the brain and is mostly used for tremor-dominant PD. If you have bilateral symptoms and complex fluctuations, DBS is still the gold standard.
In the past, surgeons did "lesioning" procedures like pallidotomies. These are permanent and irreversible. DBS is much preferred today because it's adjustable. If the settings aren't right, the doctor just changes the code on the pulse generator. If a lesion is misplaced, you can't "undo" it.
Next Steps: Navigating the Pathway
If you think you're a candidate, don't wait until you're "too far gone." The Parkinson's Foundation has noted that many people are referred for screening too late. The ideal time to evaluate is when medications are still working but starting to cause problematic fluctuations.
- Consult a Movement Disorder Specialist: Not all neurologists specialize in Parkinson's. You need a specialist who deals with DBS daily.
- Get a Neuropsychological Exam: This 4-6 hour battery of tests determines if your cognitive health is stable enough for surgery.
- Schedule a 3T MRI: Precise targeting requires high-resolution imaging to map your specific brain anatomy.
- Multidisciplinary Review: Your surgeon, neurologist, and psychologist should all agree on the plan.
Does DBS stop Parkinson's from getting worse?
No. DBS manages symptoms like tremors, stiffness, and dyskinesia, but it does not slow down or stop the underlying progression of the disease. It improves the quality of life, but the disease continues to evolve.
How much does DBS cost?
In the US, costs can range from $50,000 to $100,000. However, Medicare and most private insurance plans cover it for Parkinson's patients who meet the clinical eligibility criteria.
Will I have to have more surgeries later?
If you have a non-rechargeable system, the battery (IPG) will eventually run out and need replacement, usually every 3-5 years. Rechargeable systems can last 9-15 years, significantly reducing the number of follow-up surgeries.
Can I still take my medication after DBS?
Yes. In fact, most patients continue taking medication, but often at a much lower dose. The goal is to find a balance between the stimulation and the drugs to minimize side effects.
What are the risks of the surgery?
The main risks include a 1-3% chance of intracranial hemorrhage (bleeding in the brain) and a 5-15% risk of hardware-related complications, such as the lead shifting or an infection at the site of the implant.

william wang
April 17, 2026 AT 21:34The part about the levodopa response is a huge point for anyone considering this. It's basically the litmus test for whether the surgery will actually do anything for your specific motor symptoms. I've seen a few cases where people pushed for it despite a poor response and ended up really disappointed because the electrical stimulation just doesn't replace the chemistry if the brain isn't reacting to the dopamine in the first place.
Anmol Garg
April 18, 2026 AT 14:43It's kind of wild to think about how we're basically hacking our own neural circuitry to find peace again. The balance between the STN and GPi targets really highlights how individual every journey with this disease is. We all just want a bit of stability in the chaos, right?
Tama Weinman
April 19, 2026 AT 05:02Funny how they call it "closed-loop" technology to make it sound safe, but really it's just another way for a device to monitor your brain waves in real-time. Once you let a corporation put a pulse generator under your skin, you're basically just a node in their network. Just a thought for those who think they're "reclaiming independence" by becoming a cyborg.
Anna BB
April 19, 2026 AT 10:50The idea of a "sweet spot" in brain programming is just so poetic...!! It really shows that healing isn't a straight line, but a series of tiny adjustments...!!! I love that there is a way to bring back fluid movement to a body that feels like it's fighting itself...!!!
Maggie Graziano
April 21, 2026 AT 10:48they want us on batteries now control the signals control the mind easy way to track every move
Josephine Wyburn
April 23, 2026 AT 09:07Oh my gosh, I just cannot even imagine the absolute horror of having to spend six to twelve months just trying to find the right setting for a machine in your head, like can you actually imagine the emotional toll of that waiting period where you're just hoping and praying that the next appointment is the one that finally fixes everything 😭 and then there's the fear of infection which is just a total nightmare scenario for anyone who's already struggling with their health 😱 it's just so overwhelming and heartbreaking to think about the vulnerability of it all while you're just trying to be a normal human being again 💔
Theresa Griffin MEP
April 24, 2026 AT 08:39Medical precision is paramount. The 3T MRI is an absolute necessity.
Kim Hyunsoo
April 25, 2026 AT 23:55The way the directional leads can "steer" electricity is some seriously fancy wizardry (⊙︿⊙) I wonder if the sensation of that real-time adjustment feels like a sudden wave of clarity or just a subtle shift in how the body responds.
Cheryl C
April 27, 2026 AT 17:08USA medicine is just the best 🇺🇸 no one else does brain surgery like we do!! get those leads in there and get back to livin!! 🤠💥
Nikki Grote
April 27, 2026 AT 21:37Regarding the GPi target, it's specifically advantageous for those with severe levodopa-induced dyskinesia because it modulates the output of the basal ganglia more broadly. From a clinical perspective, the preservation of cognitive function is a significant priority, especially since the STN target can occasionally trigger transient speech issues or a perceived slowing in mental processing due to the proximity of the limbic and associative territories.
Heer Malhotra
April 28, 2026 AT 06:01It is a matter of grave concern that such expensive procedures are discussed without addressing the disparities in global healthcare access. While the West prides itself on these technological marvels, the ethical implication of a $100,000 price tag ensures that only a privileged few benefit, which is a moral failing of the current system.
Colleen Tankard
April 29, 2026 AT 23:23Seeing people get their lives back from the "off" periods is just amazing 🌟 it's like giving them a second chance at their hobbies and family time ✨
Rock Stone
April 30, 2026 AT 20:49Keep pushing forward everyone! If you're in that window where meds are starting to glitch, just get the consultation. Better to know your options now than to wish you'd started three years ago. You've got this!