When you hear the word "generic," you probably think of cheap pills that work just like the brand-name version. But when it comes to biologic drugs-like those used for rheumatoid arthritis, cancer, or psoriasis-things are not that simple. Biosimilars are not generics. They’re something else entirely. And they’re starting to change how much patients and healthcare systems pay for life-changing treatments.
What Exactly Is a Biosimilar?
A biosimilar is a biologic drug that is highly similar to an already approved biologic, called the reference product. These aren’t chemical copies like generic pills. Biologics are made from living cells-yeast, bacteria, or animal cells-and are incredibly complex. Think of it like cloning a tree. You can get a very close copy, but it’s never going to be *exactly* the same tree. That’s why biosimilars aren’t called "generics." They’re called biosimilars because they’re extremely close, with no meaningful difference in safety or effectiveness. The first biosimilar was approved in the U.S. in 2015. Since then, more have come online. By 2025, there were 10 FDA-approved biosimilars for Humira (adalimumab), one of the most prescribed and expensive biologics in history. The same pattern is happening with Stelara (ustekinumab), Enbrel, and others.How Much Do Biosimilars Actually Save?
The savings aren’t as dramatic as with traditional generics, but they’re still huge. Generics can cut prices by 80-90%. Biosimilars? They typically save 15-35% off the list price of the original biologic. But that’s just the start. For Humira, when biosimilars launched in 2023, some offered up to 85% off the list price. That sounds amazing, right? But here’s the catch: list price isn’t what most people pay. The real cost is hidden behind rebates, discounts, and complicated contracts between drugmakers, pharmacies, and insurers. Still, even after all that, biosimilars are consistently cheaper. A 2025 analysis by CSRxP found that patients paid 23% less out of pocket for biosimilars compared to the original biologic. And it’s not just about individual patients. Employers are seeing real savings. One study found that if all employees switched from a biologic to its biosimilar, an employer could save an average of $1.53 million per year. Across all self-insured U.S. companies, switching just two biologics to biosimilars could save $1.4 billion annually. Cumulative savings since 2015? Over $56 billion in the U.S. alone. In 2024, biosimilars saved the system $20.2 billion. That’s money that didn’t go to drugmakers. It went back into the system-funding other treatments, lowering premiums, or keeping care affordable.Why Don’t Biosimilars Save More?
You’d think with 10 biosimilars for Humira, the price would crash. But it hasn’t. Why? First, manufacturing is hard. Biologics require living cell cultures, sterile environments, and months of testing. A biosimilar can take 8-10 years and over $100 million to develop. That’s not like making a generic pill in a lab. Second, the market is rigged. Originator companies-like AbbVie with Humira-use rebate deals to keep their drugs on formularies. Even if a biosimilar is cheaper, the original drug might offer a bigger rebate to the pharmacy benefit manager (PBM), making the net cost look similar. This is called the "rebate trap." It keeps patients on expensive drugs even when cheaper, equally safe options exist. Third, doctors and patients are still cautious. Many assume biosimilars are "lesser." But studies show no difference in safety or effectiveness. A 2024 JAMA Network Open review confirmed that biosimilar prices kept dropping over time after they entered the market, and patients had no more side effects than those on the original.
Where Are the Biggest Savings Happening?
The U.S. lags behind Europe. In countries like Norway, Germany, and Sweden, biosimilars have captured over 80% of the market for some biologics within three years. In the U.S., originator biologics still made up 98.9% of all biologic spending in 2023. That means less than 1.1% of spending went to biosimilars-even though they’ve been around for a decade. Why the gap? Europe has stricter price controls. The U.S. doesn’t. European governments negotiate prices directly. In the U.S., pricing is a mess of private contracts. Take Stelara. As of July 2025, nine biosimilars hit the market with prices as low as 90% below the original list price. But if the PBM is getting a 70% rebate on the original, the patient might still end up paying more for the biosimilar because it’s not on their formulary.What’s Blocking the Next Wave of Savings?
Here’s the scary part: 118 biologics are expected to lose patent protection over the next 10 years. But only 12 of them have biosimilars in development. That means 90% of upcoming biologics will have no competition. No biosimilars. No price drops. No savings. That’s a $234 billion opportunity lost. The European Union has biosimilars in development for 73% of high-sales biologics. The U.S.? Only 23%. We’re not just falling behind-we’re ignoring the problem. The FDA has made steps to speed up approvals. The Inflation Reduction Act of 2022 gave Medicare more power to negotiate prices. But without policy changes-like requiring biosimilar preference in Medicare Part D or banning rebate-based formulary steering-those savings won’t happen.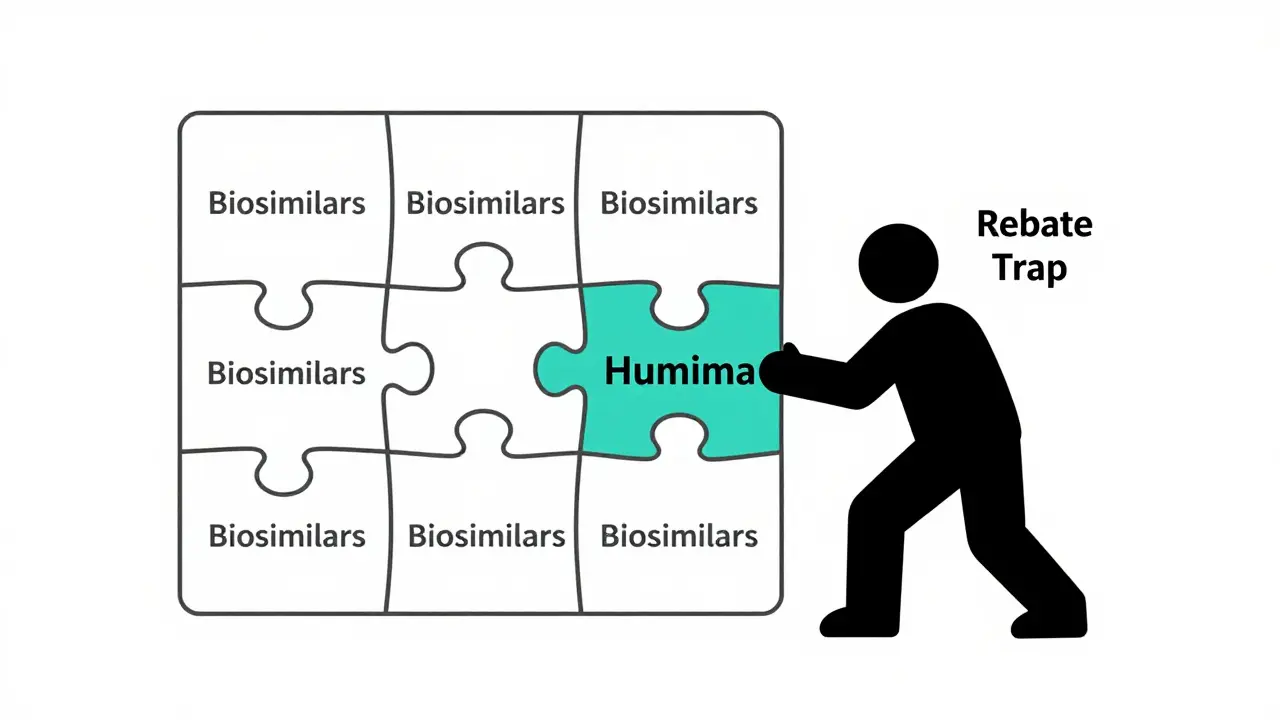
How Can Patients and Employers Get More Savings?
If you’re a patient, ask your doctor: "Is there a biosimilar for my drug?" Many don’t know. If you’re on Humira, there are 10 biosimilar options. Switching is safe, and your out-of-pocket cost could drop by 20-45%. Employers and health plans can do more:- Place biosimilars as the preferred option on formularies
- Require patients to try the biosimilar before approving the original (step therapy)
- Negotiate contracts that reward biosimilar use, not rebates
- Use data tools like Segal’s SHAPE to track spending and identify where savings are being lost
- Educate doctors and patients-misinformation is the biggest barrier


Laura Gabel
March 17, 2026 AT 12:45Prathamesh Ghodke
March 18, 2026 AT 07:35jerome Reverdy
March 19, 2026 AT 16:13Andrew Mamone
March 19, 2026 AT 21:02MALYN RICABLANCA
March 19, 2026 AT 22:47gemeika hernandez
March 21, 2026 AT 06:21Nicole Blain
March 22, 2026 AT 18:22Kathy Underhill
March 24, 2026 AT 06:15Sanjana Rajan
March 25, 2026 AT 14:45Kyle Young
March 26, 2026 AT 21:54Aileen Nasywa Shabira
March 27, 2026 AT 04:16Ryan Voeltner
March 27, 2026 AT 11:55Linda Olsson
March 27, 2026 AT 12:35