When you pick up a prescription, you might not realize that the pill you’re holding could be a different brand than what your doctor wrote on the script. That’s because of generic substitution laws-rules that let pharmacists swap out brand-name drugs for cheaper generic versions. But here’s the catch: these rules aren’t the same across the U.S. Every state has its own version, and some are strict, others are loose, and a few even require your explicit permission before making the switch.
How Generic Substitution Works (and Why It Matters)
Generic drugs contain the same active ingredients as brand-name drugs, work the same way, and are held to the same safety standards by the FDA. The only differences are usually in color, shape, or inactive ingredients like fillers. The FDA’s Orange Book is the official federal list that identifies which generics are considered therapeutically equivalent to their brand-name counterparts. This is the foundation for substitution laws.
Why does this matter? Cost. Generic drugs typically save patients 80% or more compared to brand-name versions. In 2022, generics made up 90.7% of all prescriptions filled in the U.S., saving consumers an average of $313 per prescription. Without substitution laws, many people would pay more-sometimes hundreds of dollars extra-just because a doctor wrote the brand name.
State Laws: Mandatory vs. Permissive Substitution
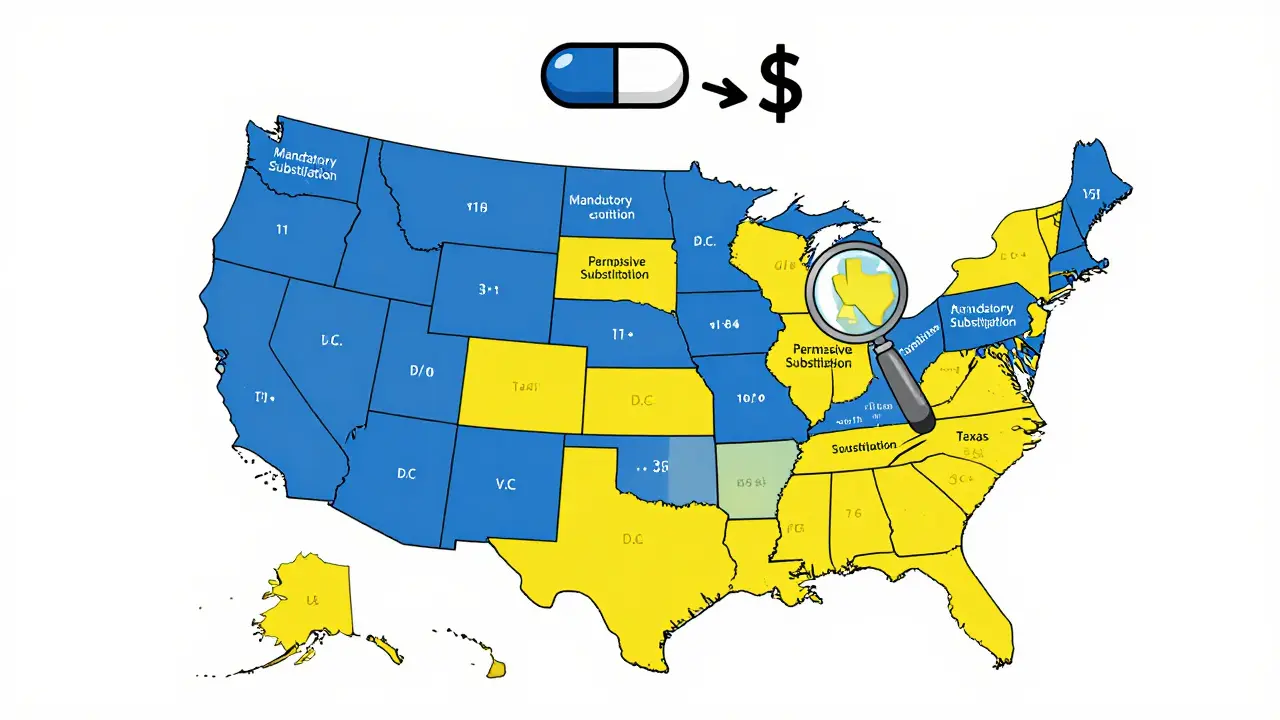
Not all states treat substitution the same. There’s a clear split between states that force pharmacists to substitute and those that leave it up to them.
- 19 states require substitution when a generic is available. This includes big ones like California, New York, and Texas. In these places, if a generic is FDA-approved and available, the pharmacist must dispense it unless the doctor says "do not substitute" on the prescription.
- 31 states and Washington, D.C. allow substitution but don’t require it. Pharmacists here can choose whether to swap the drug, but they usually do-especially if the patient saves money. Some of these states, like Alaska, Delaware, Maine, and New Hampshire, require pharmacies to post signs telling patients substitution is possible.
This difference isn’t just paperwork. Studies show that states with mandatory substitution laws see generic use rates 8-12 percentage points higher than permissive states. That means more people are getting affordable meds where substitution is required.
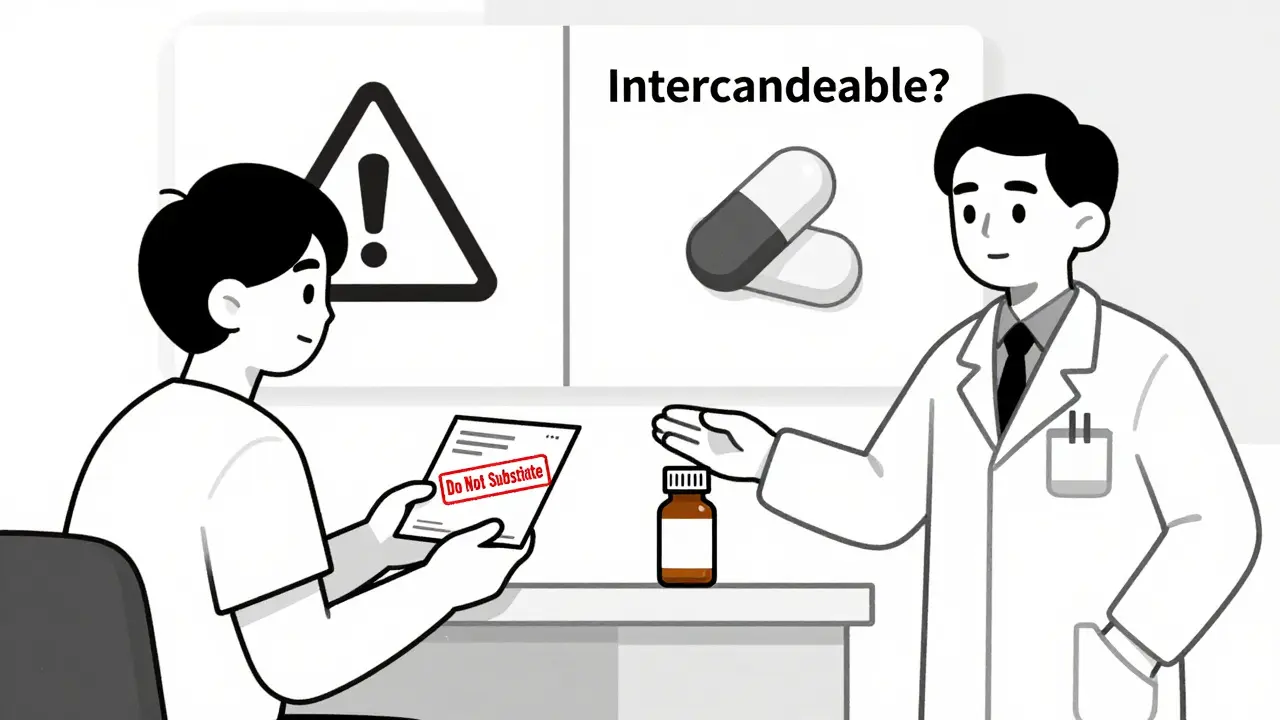
Patient Consent: Do You Have to Say Yes?
Here’s where it gets personal. Some states think you should have a say before your medication changes. Others believe notification is enough.
- 7 states plus Washington, D.C. require explicit patient consent before substitution can happen. These include Connecticut, Hawaii, Maine, Maryland, New Hampshire, Vermont, and West Virginia. In these places, the pharmacist must get your verbal or written okay before switching your drug.
- 31 states plus D.C. require notification after substitution. You don’t have to agree ahead of time-you just have to be told afterward. This usually happens on the prescription label or via a handout.
Why does consent matter? For some drugs, even tiny differences can cause problems. That’s why Hawaii bans substitution of antiepileptic drugs without both patient and doctor approval. The same goes for drugs like warfarin and levothyroxine, which have a narrow therapeutic index-meaning the difference between a helpful dose and a harmful one is very small.
Formularies: What’s Allowed and What’s Not
Some states don’t just say "yes" or "no" to substitution-they create lists.
- Positive formularies list which generics can be substituted. Kentucky, for example, keeps a list of drugs that are not allowed to be swapped, including antiepileptic drugs and digitalis glycosides. Pharmacists in Kentucky can’t substitute those, no matter what.
- Negative formularies list which drugs can’t be substituted. Most states use this approach. For example, Oklahoma bans substitution unless the prescriber or patient gives permission. That means the default is "no substitution" unless someone says otherwise.
This creates a patchwork. A pharmacist in Texas might swap your blood pressure med without a second thought. But if you cross into Oklahoma with the same script, they can’t touch it unless your doctor signed off.
Biosimilars: The New Frontier
Biosimilars are the generic version of biologic drugs-complex medications used for conditions like rheumatoid arthritis, cancer, and psoriasis. They’re harder to copy than small-molecule generics, so the rules are stricter.
- 45 states have tougher rules for biosimilars than for regular generics.
- 9 states (like Alabama, Arizona, and Massachusetts) require patient notification for biosimilar substitution but not for regular generics.
- Only 11.2% of biologic prescriptions in 2023 were filled with biosimilars-partly because of confusing state laws.
- All states that allow biosimilar substitution require the FDA to label the product as "interchangeable." That’s a higher bar than for regular generics.
Patients often don’t realize they’ve been switched. A 2022 survey by the National Psoriasis Foundation found that 42% of people on biologics didn’t know their drug had been changed to a biosimilar-even though state laws required notification.
Liability: Who Gets Blamed if Something Goes Wrong?
Pharmacists aren’t just following rules-they’re worried about lawsuits.
Twenty-four states-including Alabama, Missouri, and Oregon-don’t give pharmacists legal protection if a substituted drug causes harm. That means if a patient has a bad reaction after a generic swap, the pharmacist could be held liable.
As a result, many pharmacists in those states avoid substitution altogether, especially for high-risk drugs. One pharmacist told the National Community Pharmacists Association they spend 15-30 minutes a day just checking state rules. In border towns, where patients live in one state and fill prescriptions in another, it’s even worse.
What’s Changing? The Push for Standardization
The current system is messy. A pharmacist in Ohio might be required to substitute, but if they work in Pennsylvania, they can’t unless the patient agrees. This confusion costs time, money, and sometimes safety.
Between 2018 and 2022, states that simplified their laws saw generic use jump by an average of 6.8 percentage points. In states that removed consent requirements, the increase was as high as 11.2 points.
Right now, 12 states are considering the "State Harmonization of Generic Substitution Act," which would create common rules across participating states. The FDA also updated its Orange Book in 2022 to include new "interchangeability" designations for complex generics, prompting 18 states to review their laws.
But the big question remains: Will we ever have one national standard? For now, the answer is no. And until then, patients and pharmacists alike will keep navigating 51 different sets of rules.
What You Can Do
- Ask your pharmacist: "Is this a generic? Can I get it?"
- Check your prescription label. It should say if a substitution occurred.
- If you take a drug with a narrow therapeutic index (like warfarin, levothyroxine, or seizure meds), ask your doctor to write "dispense as written" or "no substitution."
- Know your state’s rules. The National Association of Boards of Pharmacy has an interactive map that shows your state’s current laws.
Generic substitution saves money. But it only works if you know how it works-and whether you have the right to say no.
Can a pharmacist substitute my brand-name drug without telling me?
In 31 states and Washington, D.C., pharmacists are required to notify you after substitution, usually on the prescription label. But in 7 states plus D.C., they must get your explicit consent before swapping the drug. If you’re in a state that requires consent, they can’t substitute without your okay. Always check your prescription label and ask if you’re unsure.
Are all generics safe to substitute?
The FDA approves generics as therapeutically equivalent to brand-name drugs, but some medications have a narrow therapeutic index-meaning small changes in dosage can cause big effects. Drugs like warfarin, levothyroxine, and certain seizure medications fall into this category. Some states ban substitution for these drugs entirely. If you take one of these, ask your doctor to write "dispense as written" on your prescription.
Why do some states require patient consent for substitution?
States that require consent do so because of concerns over patient safety, especially with drugs where even small changes in absorption or metabolism can cause harm. The American Epilepsy Society and other patient groups pushed for these rules after reports of seizures in patients switched to different generic versions of antiepileptic drugs. While studies show most generics are safe, these states err on the side of caution.
Do biosimilars follow the same rules as regular generics?
No. Biosimilars are more complex and require FDA designation as "interchangeable" before substitution is allowed. Forty-five states have stricter rules for biosimilars than for regular generics. Some states require prescriber notification, patient consent, or both. As of 2023, only 11.2% of biologic prescriptions were filled with biosimilars, partly because of these confusing and inconsistent laws.
Can I request a brand-name drug even if a generic is available?
Yes. You can always ask your pharmacist for the brand-name version. In states with mandatory substitution, the pharmacist must still give you the brand if you request it-though you may have to pay more out of pocket. If your doctor wrote "dispense as written," that overrides any substitution rules. Always communicate your preference clearly.

kirti juneja
February 27, 2026 AT 14:12Wow, this is wild! I never realized how much chaos there is across states-like, one day you’re in Texas getting a cheap generic, next day you’re in Oklahoma and the pharmacist just stares at you like you asked for a unicorn. I’m from India, and here we just get the generic unless it’s super niche. No drama, no paperwork. Why can’t the US just standardize this? Save time, save cash, save lives.
Gabrielle Conroy
February 28, 2026 AT 20:11This is so important!! I work in a pharmacy, and honestly? I hate the inconsistency!! One day I’m explaining to a patient why we can’t swap their levothyroxine in Maine, next day I’m doing it automatically in California. It’s exhausting!! And don’t even get me started on biosimilars-patients think they’re ‘fake drugs’ because they’re not told properly!! We need a national standard, like, yesterday!!! 🙏😭
Spenser Bickett
March 1, 2026 AT 00:14Nick Hamby
March 1, 2026 AT 16:15What fascinates me about this system is how it reveals the tension between efficiency and autonomy. On one hand, mandatory substitution saves billions and democratizes access. On the other, the consent requirements reflect a deeply held belief: that the body is not a commodity to be optimized. There’s wisdom in both. But perhaps we’ve overcorrected-by making consent a legal hurdle rather than a dialogue. What if pharmacists were trained to have brief, empathetic conversations about therapeutic equivalence? Not forms. Not notices. Conversation. That’s the missing layer.
Steven Pam
March 1, 2026 AT 22:16Love this breakdown! Seriously, I had no idea biosimilars had their own whole rulebook. I’m on a biologic for psoriasis and had no clue mine got swapped last year. My doctor didn’t say anything, the label didn’t flag it, and I just thought I was getting ‘the same thing.’ Now I always ask. And yeah, I’m gonna check my state’s rules tonight. Knowledge is power, folks! 💪💊
Shalini Gautam
March 3, 2026 AT 01:25Haley Gumm
March 4, 2026 AT 16:42Let’s be real: the consent laws are performative. They make people feel safe, but they don’t improve outcomes. Studies show no significant difference in adverse events between consent and notification states. Meanwhile, patients in consent states delay refills, skip meds, or pay out of pocket because they don’t understand the process. It’s not protection-it’s bureaucratic theater. And don’t even get me started on pharmacists who avoid substitution out of fear. That’s not safety. That’s negligence dressed up as caution.
Christopher Wiedenhaupt
March 4, 2026 AT 17:46John Smith
March 6, 2026 AT 12:43Natanya Green
March 6, 2026 AT 17:05