The FDA Orange Book is the go-to source for anyone trying to figure out when a brand-name drug’s patent protection ends and generics can legally hit the market. If you’re a pharmacist, a generic drug manufacturer, or even a patient wondering why a cheaper version isn’t available yet, knowing where to look in the Orange Book can save you time, money, and confusion. But it’s not as simple as just typing in a drug name and getting a clear answer. The system has layers, quirks, and gaps - and if you don’t know what to look for, you might miss the real expiration date entirely.
What the FDA Orange Book Actually Is
The official name is Approved Drug Products with Therapeutic Equivalence Evaluations, but everyone calls it the Orange Book. It started in 1985 after Congress passed the Hatch-Waxman Act, which aimed to balance drug innovation with affordable access. The idea was simple: let brand-name companies keep patent protection, but give generic makers a clear path to enter the market once those patents expire.
It’s not just a list of drugs. The Orange Book tracks every approved small-molecule drug in the U.S. - including the patent numbers, their expiration dates, and any regulatory exclusivity periods. These aren’t just random dates. They’re legally binding deadlines that determine when generic versions can be approved and sold. Missing one of these dates can mean losing millions in revenue or delaying a drug launch by months.
Since 2008, the Orange Book has been entirely digital. The FDA updates it daily, and you can download the full dataset for bulk analysis. But most people use the web interface, which is free and publicly accessible.
How to Find Patent Expiration Dates Online
To find a patent expiration date, go to the Electronic Orange Book. You can search by:
- Brand name (like Brilinta or Lipitor)
- Generic name (like ticagrelor or atorvastatin)
- Application number (the FDA’s internal ID for each drug)
- Company name (the sponsor, like AstraZeneca or Pfizer)
Once you find the drug, click on the Application Number link. That takes you to a detailed page. Scroll down and click View under the Patents section. Here’s where you’ll see the actual expiration dates - listed as MMM DD, YYYY, like July 9, 2021.
Don’t just look at the first patent. Many drugs have multiple patents covering different things: the active ingredient, how it’s made, how it’s used, or even how it’s packaged. Each one has its own expiration date. The last one to expire is the real deadline for generic entry.
What the Dates Really Mean - and What They Don’t
The expiration date you see isn’t just the patent’s original end date. It includes Patent Term Extensions (PTE), which add time back to the patent because of delays in FDA approval. A drug that was supposed to expire in 2020 might now show 2025 because it took five years to get approved.
But here’s the catch: the Orange Book doesn’t always reflect early expirations. If a patent owner stops paying maintenance fees, the patent dies early - but the Orange Book doesn’t automatically remove it. A 2023 study found that 46% of patents listed in the Orange Book actually expired earlier than what’s shown. That’s not a glitch - it’s a known gap. The FDA doesn’t retroactively update old listings.
So if you’re planning to launch a generic drug, you can’t rely on the Orange Book alone. You need to cross-check with the USPTO Patent Center. That’s where you can see if maintenance fees were paid. If they weren’t, the patent is dead - even if the Orange Book says otherwise.
Pediatric Exclusivity: The Hidden 6-Month Extension
Here’s where things get confusing. If a drug maker conducts pediatric studies, the FDA gives them an extra six months of market protection. This isn’t a new patent. It’s an extension tacked onto existing patents and exclusivity periods.
In the Orange Book, this shows up as two entries for the same patent:
- One with the original expiration date
- One with the original date + six months
For example, if a patent expires on January 1, 2024, and pediatric exclusivity applies, you’ll see two lines:
- Patent Expiration: January 1, 2024
- Patent Expiration: July 1, 2024
The second date is the real one. Generic companies can’t launch until that later date. If you miss this, you’ll think the market is open when it’s not.
Patent Use Codes - The Key to Understanding What’s Covered
Each patent in the Orange Book has a Patent Use Code - a code like U-123 or U-456. These codes tell you what the patent actually protects. Is it for treating high blood pressure? Or for a specific dosage form? Or for a new way to administer the drug?
Not all patents block generics. Some only protect one use of the drug. If a generic company can prove their version only treats a different condition, they might launch even while the patent is still active.
The FDA has a Patent Use Code search tool to decode these. But it’s clunky. Many users report it doesn’t load properly. If you’re serious about this, save the codes and look them up in the FDA’s official documentation - it’s more reliable.
Delisting: When a Patent Disappears
Look for the Patent Delist Request Flag. If it says “Y,” the patent owner asked the FDA to remove it. That usually means one of two things:
- The patent was invalidated in court
- The company decided it’s no longer worth defending
Either way, this is a green flag for generic manufacturers. If a patent is delisted, it’s no longer a legal barrier. The FDA doesn’t remove it immediately - it stays visible with the “Y” flag - but it’s no longer enforceable.
How to Get the Full Data (For Developers and Analysts)
If you’re doing serious research, the web interface won’t cut it. Download the Orange Book Data Files. They’re updated daily and come as CSV or Excel files. Key columns include:
- Product No - the FDA’s internal product ID
- Patent No - the actual patent number from the USPTO
- Patent Expiration - the date in MM/DD/YYYY format
- Drug Substance Flag - “Y” if the patent covers the active ingredient
- Drug Product Flag - “Y” if it covers the specific formulation
- Patent Use Code - the U-code
- Delist Requested Flag - “Y” if delisted
With this data, you can build your own tools to track expirations across hundreds of drugs. Many generic companies use automated scripts to monitor these files daily.
What You Shouldn’t Trust
- Third-party databases - Many sites scrape the Orange Book but don’t update daily. Some are months out of date.
- Patent expiration calculators - They often ignore PTE and pediatric exclusivity.
- Old printed copies - The last printed version was in 1999. Everything since is digital.
- Assuming all patents are equal - A patent on a pill coating doesn’t block a generic that uses a different coating.
Real-World Impact
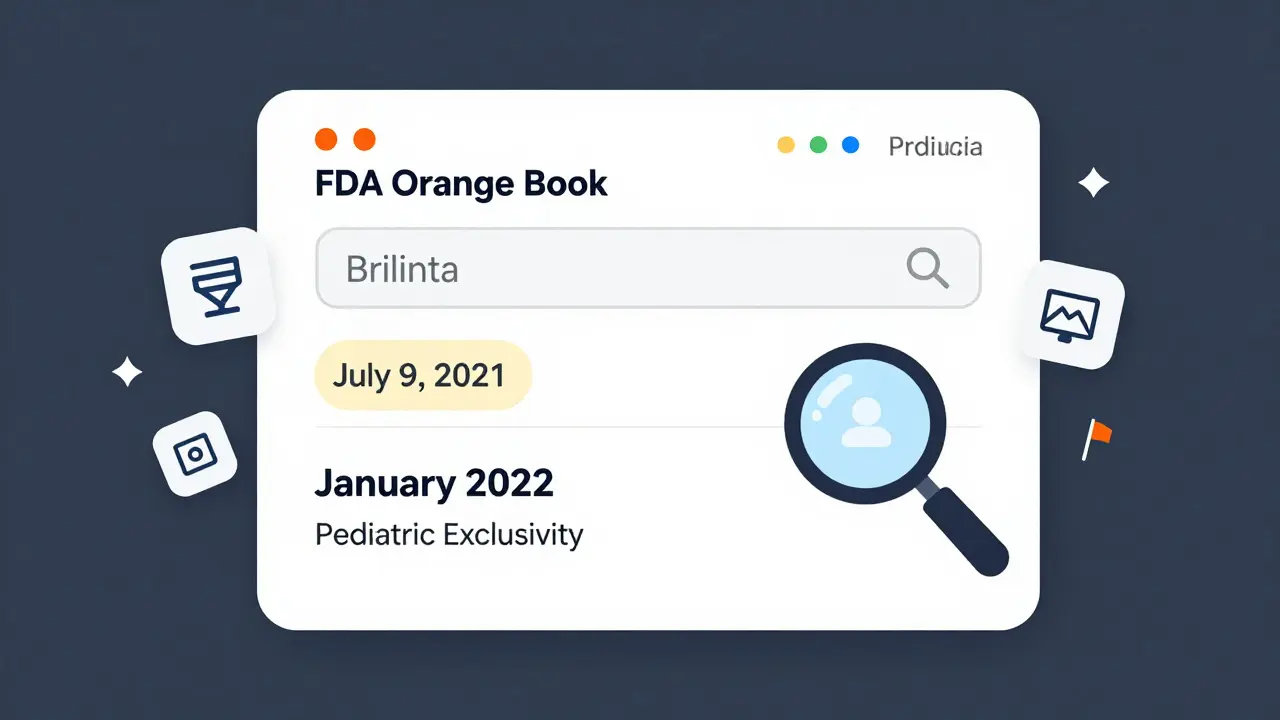
In 2023, the drug Brilinta (ticagrelor) had multiple patents listed. The last one expired on July 9, 2021. But because of pediatric exclusivity, the real date was July 20, 2016 - wait, no. That’s wrong. Actually, the exclusivity extended the protection. The original patent expired in 2021, but the exclusivity added six months, so generics couldn’t launch until January 2022. If you only looked at the first date, you’d have been wrong.
That’s why experts say: Always check the latest version. Always cross-reference with USPTO. Always look for delistings.
The FDA says over 78% of brand-name drug revenue will face generic competition by 2025. That means more drugs are hitting patent cliffs. The Orange Book isn’t perfect - but if you know how to read it, it’s still the most reliable tool you have.
Where can I find the official FDA Orange Book online?
The official Electronic Orange Book is available at https://www.accessdata.fda.gov/scripts/cder/ob/index.cfm. This is the only source updated daily by the FDA. Avoid third-party sites - they often lag behind.
Do all patents listed in the Orange Book block generic drugs?
No. Only patents that cover the drug’s active ingredient or its approved use can block generics. Some patents cover delivery methods, packaging, or minor formulations - and generics can still launch if they avoid those. Always check the Patent Use Code to see what’s actually protected.
Why does the Orange Book show two expiration dates for the same patent?
That’s pediatric exclusivity. When a drug maker studies the drug in children, the FDA adds six months of protection to all existing patents. The Orange Book shows both the original date and the extended date. The later one is the real deadline for generic entry.
Can a patent expire early even if the Orange Book says otherwise?
Yes. If the patent owner doesn’t pay maintenance fees, the patent dies - but the Orange Book doesn’t remove it. Studies show nearly half of listed patents expire early. Always verify with the USPTO Patent Center to check payment status.
What’s the difference between patent expiration and exclusivity?
Patents are granted by the USPTO and protect inventions - like the chemical structure or a new use. Exclusivity is granted by the FDA and protects against competition for a set time after approval, regardless of patents. A drug can have one, both, or neither. Exclusivity can extend beyond patent life.
Is the Orange Book updated in real time?
The web interface updates daily, and the downloadable data files are refreshed every day. But changes take 24-48 hours to appear. If a patent is delisted or extended on Monday, it won’t show up until Tuesday or Wednesday.
Do biologics show up in the Orange Book?
No. The Orange Book only covers small-molecule drugs - the kind you take as pills. Biologics (like insulin, vaccines, or monoclonal antibodies) are listed in a separate FDA database called the Purple Book.
Can I trust the expiration dates if I’m planning to launch a generic drug?
Use the Orange Book as your starting point, but don’t rely on it alone. Always verify patent status through the USPTO Patent Center, check for delistings, and confirm whether pediatric exclusivity applies. Many lawsuits have been filed because companies assumed the Orange Book was complete - and they were wrong.



Tom Bolt
March 11, 2026 AT 07:37The FDA Orange Book is a masterpiece of bureaucratic elegance-every patent number, every use code, every delisting flag meticulously documented. And yet, somehow, we still have pharmacists missing expiration dates because they didn’t cross-reference with the USPTO. The irony is delicious. This isn’t a database; it’s a legal minefield disguised as a public service.
Mike Winter
March 12, 2026 AT 22:16It's fascinating how the system tries to balance innovation and access, but the reality is messier than the theory. Patents aren't just legal instruments-they're economic weapons. And when maintenance fees lapse, it's not a glitch, it's a silent revolution in the market. The Orange Book is a map, but the terrain keeps changing beneath it.
Randall Walker
March 13, 2026 AT 04:54So let me get this straight… we’ve got a government database that’s updated daily… but it’s still wrong half the time? And we’re supposed to trust it? Yeah, I’ll just hop on over to the USPTO site, which crashes if you look at it funny, and hope my coffee doesn’t spill on the keyboard. Classic.
Donnie DeMarco
March 14, 2026 AT 13:31Yo I just spent 3 hours trying to decode one drug’s patent maze and now I feel like I’ve been through a pharmaceutical labyrinth with a blindfold. The Orange Book? More like the Orange Confusion. But hey, at least it’s free. Unlike my sanity.
Shourya Tanay
March 15, 2026 AT 00:27The interplay between patent term extension (PTE) and pediatric exclusivity introduces a bifurcated temporal framework that complicates market entry projections. One must parse not only the statutory language but also the administrative interpretations embedded in the use codes, which are often ambiguously indexed. The data architecture, while comprehensive, lacks semantic interoperability with external regulatory repositories.
LiV Beau
March 16, 2026 AT 14:19OMG I just learned about the pediatric exclusivity thing and I’m obsessed 😍 This is like a secret superpower the pharma companies get but no one talks about! I’m gonna start a TikTok series called "Patent Secrets the FDA Doesn’t Want You to Know" 📚💊 #GenericDrugHacks
Adam Kleinberg
March 17, 2026 AT 03:53Of course the government database is outdated. It’s all part of the grand scheme to keep generics expensive so the pharmaceutical elite can keep siphoning billions from the working class. They don’t want you to know about the USPTO-because then you’d realize how easily patents are manipulated. Wake up.
Gene Forte
March 17, 2026 AT 11:00The Orange Book represents one of the most important public health tools we have. It is not perfect, but it is transparent, accessible, and continuously updated. With diligence and cross-verification, it empowers manufacturers, pharmacists, and patients alike. We should honor its intent, not dismiss its utility.
David L. Thomas
March 19, 2026 AT 07:40Honestly? I just download the CSV and run a quick script. The web UI is a joke. If you're doing this manually, you're doing it wrong. Automation is the only sane way to handle this. Also, pediatric exclusivity? That’s the real game-changer. Most people miss it because they’re too busy staring at the first expiration date.
Bridgette Pulliam
March 20, 2026 AT 18:29I appreciate how detailed this is. I’m a community pharmacist, and I’ve seen too many patients confused because they thought a generic was available when it wasn’t. This kind of clarity matters. Thank you for taking the time to explain the nuances. It’s easy to overlook the little things-like delisting flags-but they change everything.
Miranda Varn-Harper
March 22, 2026 AT 08:17Actually, I think the Orange Book is a well-intentioned failure. The system is designed to confuse. Why else would they make the patent use codes so cryptic? And don’t get me started on the "Y" flags-why not just say "DELETED"? This feels intentional. Like someone wants generics to fail.
Alexander Erb
March 23, 2026 AT 14:09Bro I just used this for my startup and it saved us like $2M in legal fees 🤯 The key is: always check the USPTO. The Orange Book is your buddy, not your boss. And yeah, pediatric exclusivity? Total curveball. I missed it once and almost got sued. Don’t be me.
Denise Jordan
March 25, 2026 AT 06:40So… you’re telling me I have to check TWO websites? And read legal jargon? And look at CSV files? Ugh. Can’t we just have an app that tells us when the generics drop? I’m tired.
Kenneth Zieden-Weber
March 26, 2026 AT 14:16You know what’s wild? The fact that the FDA updates this daily but still doesn’t auto-remove expired patents. It’s like they’re leaving breadcrumbs for people who care enough to follow them. That’s not incompetence-that’s a filter. If you’re not digging deep, you’re not meant to be in this game. Welcome to pharma.